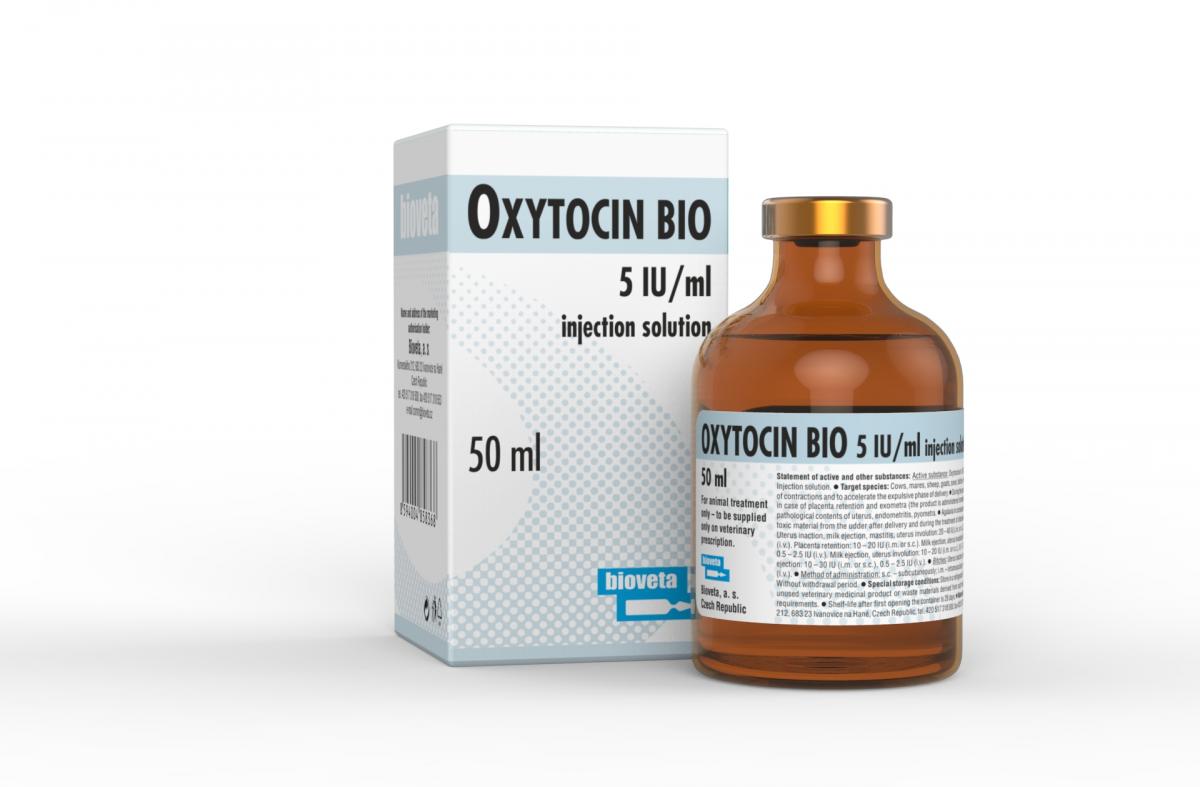
OXYTOCIN BIO 5 IU/ml injection solution
Homepage Products Veterinary products OXYTOCIN BIO 5 IU/ml injection solution
Hormonal product containing oxytocin.
| type of preparative: | Hormones |
|---|---|
| target species animals: | Cattle, Dog, Goat, Horse, Pig, Sheep |
Pharmaceutical form
Injection solution.
Clear colourless solution, without visible particles.
Composition
Oxytocinum 5.0 IU
Indication
To support delivery in case of primary and secondary depression of contractions and to accelerate the expulsive phase of delivery.
During the puerperal period: depression of uterine muscle contractility: To stimulate involution in case of placenta retention and exometra (the product is administered immediately after delivery or caesarotomy and two to four hours later), to remove the pathological contents of uterus, endometritis, pyometra.
Agalaxia in consequence of milk production disorder in all target species. To remove residual milk and toxic material from the udder after delivery and during the treatment of infectious mastitis in cows.
Target animals
Cows, mares, sheep, goats, sows, bitches.
Dosage and application
Cows
Uterus inaction, milk ejection, mastitis, uterus involution: 20– 40 IU (i.m. or s.c.), 2.5 – 10 IU (i.v.).
Mares
Uterus inaction: 20 – 40 IU (i.m. or s.c.), 2.5 – 10 IU (i.v.).
Placenta retention: 10 – 20 IU (i.m. or s.c.).
Milk ejection, uterus involution: 40 IU (i.m. or s.c.), 10 IU (i.v.).
Sheep, goats
Uterus inaction: 10 IU (i.m. or s.c.), 0.5 – 2.5 IU (i.v.).
Milk ejection, uterus involution: 10 – 20 IU (i.m. or s.c.), 0.5 – 2.5 IU (i.v.).
Sows
Uterus inaction, uterus involution, placenta retention, milk ejection: 10 – 30 IU (i.m. or s.c.), 0.5 – 2.5 IU (i.v.).
Bitches
Uterus inaction, uterus involution, placenta retention, milk ejection 2 – 10 IU (i.m. or s.c.), 0.5 IU (i.v.).
Method of administration
The product may be applied intramuscularly (i.m.), subcutaneously (s.c.) or intravenously (i.v.). For the intravenous infusion the product may be diluted with physiological solution or 5 % glucose solution.
In obstetric indications, a slow intravenous, subcutaneous or intramuscular application is possible. Repeated administration of small doses is preferred to one big dose. In sheep, the intramuscular application should be preferred.
The slow intravenous application is used to produce ejection of residual milk and retained placenta. For the intravenous application of small doses the respective product should be diluted in a higher amount of physiological solution.
Storage
Store in a refrigerator (2°C – 8°C).
Protect from frost.
Withdrawal period
Meat: Zero days.
Milk: Zero days.
Shelf life
The shelf life of the veterinary medicinal product in intact package is 2 years.
Shelf-life after first opening the container is 28 days.
Package
1 x 10 ml, 5 x 10 ml, 10 x 10 ml, 1 x 20 ml, 5 x 20 ml, 10 x 20 ml, 1 x 50 ml, 12 x 50 ml, 24 x 50 ml.
Not all pack sizes may be marketed.